Whereas in 2003, the first commercial gene therapy medicine ( Gendicine) was available on the market in China. In 2001, the 500th gene therapy clinical trial was submitted to the FDA/NIH for approval. A major drawback came in 1999 with the first gene therapy death (see also video 5). Soon it became clear that the biology of human gene therapy is very complex, and there are many techniques that still need to be developed and diseases that need to be understood more fully before gene therapy can be used appropriately. Gene therapy actually started around 1984 when Gluzman, Carter & Muzyczka developed a gene delivery system derived from adenoviruses and adeno-associated viruses. 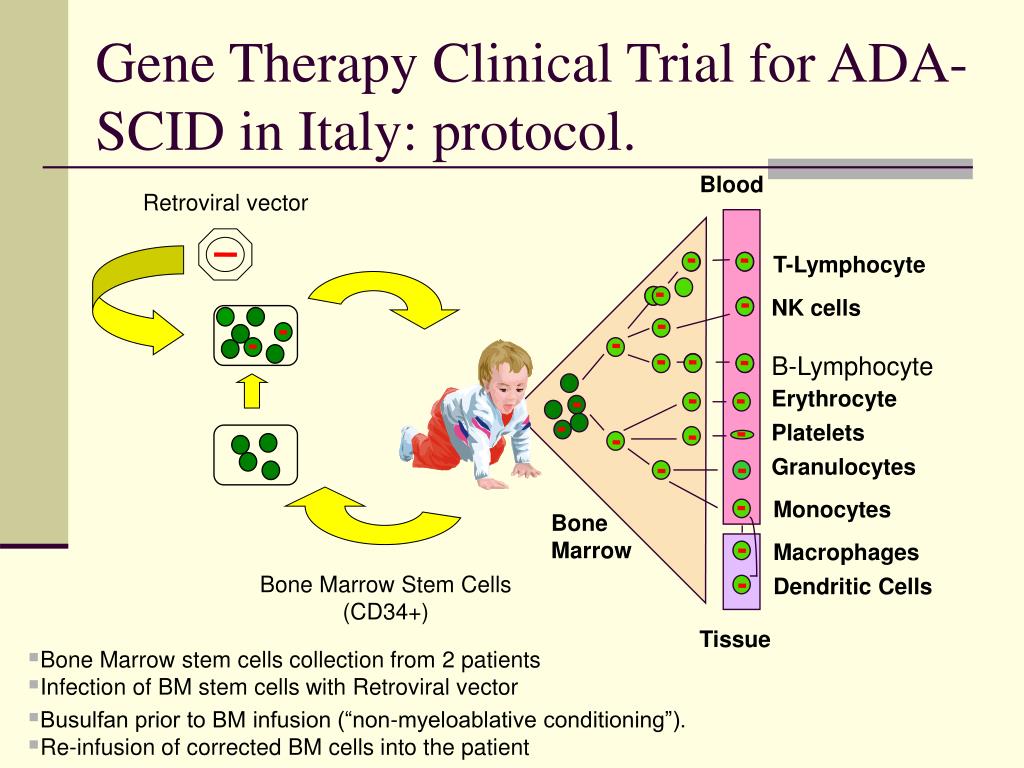
Even small amounts of the enzyme are known to be beneficial, while larger amounts are also tolerated well.Īlthough this simplified explanation of a gene therapy procedure sounds like a happy ending, it is little more than an optimistic first chapter in a long story the road to the first approved gene therapy procedure was rocky and fraught with controversy. In addition, the gene is regulated in a simple, “always-on” fashion, unlike many genes whose regulation is complex, and the amount of ADA present does not need to be precisely regulated. The reasons for selecting this disease for the first approved human clinical gene therapy trial is that the disease is caused by a defect in a single gene, which increases the likelihood that gene therapy will succeed. Video 2: Gene therapy for Muscular Dystrophy ( YouTube, 1:29) As of early 2007, she was still in good health, and she was attending college. This procedure was not a cure the white blood cells treated genetically only work for a few months, after which, the process must be repeated. Laboratory tests have shown that the therapy strengthened her immune system by 40% she no longer has recurrent colds, she has been allowed to attend school, and she was immunized against whooping cough. In her gene therapy procedure, doctors removed white blood cells from the child's body, let the cells grow in the lab, inserted the missing gene into the cells, and then infused the genetically modified blood cells back into the patient's bloodstream. She led a cloistered existence avoiding contact with people outside her family, remaining in the sterile environment of her home, and battling frequent illnesses with massive amounts of antibiotics. Children with this illness usually develop overwhelming infections and rarely survive to adulthood a common childhood illness is life-threatening. 
Born with a rare genetic disease called ( SCID), she lacked a healthy immune system, and was vulnerable to every passing germ or infection. On September 14, 1990, the first approved gene therapy procedure was performed on four-year old patient (see video 1).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
